2019
Various effects of ion implantation. — A novel method has been demonstrated in collaboration with Centre for Energy Research, to produce SiC patterns at room temperature, which is scalable up to wafer size. Two differently masked (LB film and lithographic grid) Si (20nm)/C (10nm)/Si(20nm)/C (10nm) /Si substrates have been irradiated by 120 keV, 3×1016 Xe+/cm2. Due to ion beam mixing a SiC rich layer formation on the non-covered parts of the sample can be observed, while the covered regions have not changed.
In collaboration with Atomki, Nuclear Physics Institute (Czech Republic), Institute of Applied Physics “Nello Carrara” (Italy) and University of Verona (Italy), a new method, i.e., direct writing with high-energy focused microbeam of medium-mass ions at low fluences, has been realized for the fabrication of optical channel waveguides. The lowest propagation loss in a Er3+ doped tungsten-tellurite glass waveguide, measured at a wavelength of 1400 nm after a 30 min thermal annealing at 150 °C, was 1.5 dB/cm. Due to the predominant electronic interaction, a fluence of 4.6·1014 ion/cm2, more than an order of magnitude lower than those applied in low-energy implantation, was sufficient for effective waveguide formation. The low losses of the fabricated waveguides open the use of this technique to test the fabrication of planar guided optical amplifiers and lasers in materials such as tellurite glasses where it can be difficult to use other techniques.
Radiation tolerance of organic materials has been investigated in collaboration with Atomki. Polymers have exact and known chemical structure, so they are ideal model materials to study ion beam irradiation induced chemical processes. Real-time Rutherford backscattering spectrometry and elastic recoil detection (RBS/ERDA), which was implemented to monitor the hydrogen loss caused by the He-irradiation used for the analysis itself, revealed that the hydrogen content decreased significantly. The formation of small molecule fragments and their escape from the sample was followed indirectly by RBS, simultaneously with the ERDA measurements. At lower fluences the oxygen content decreased as well. RBS/ERDA completed with infrared spectroscopy proved to be an excellent tool to study the changes of poly(methyl methacrylate) (PMMA) upon He+ irradiation, and thus to further our understanding of the complex chemical processes taking place (Fig. 1).
Advanced cultural heritage research. — Within the frame of EU IPERION CH project, non-destructive compositional analyses were performed on glass objects found in archaeological excavations from Dobroudja at the border of Black Sea. The findings were shards of late Roman glass (III-VI Centuries AD) and fragments of coloured Byzantine bracelets (X-XII Centuries AD). The analyses were done by external milli-beam PIXE (Particle Induced X-ray Emission spectrometry) with 2.5 MeV protons at Wigner RCP and by PGAA (Prompt Gamma Activation Analysis) at the cold neutron beam of the Budapest Research Reactor (Centre for Energy Research). PGAA analyses have shown that the Roman Imperial glasses are soda-lime-silica glasses using natron (a substance containing sodium salts from some lakes in Northern
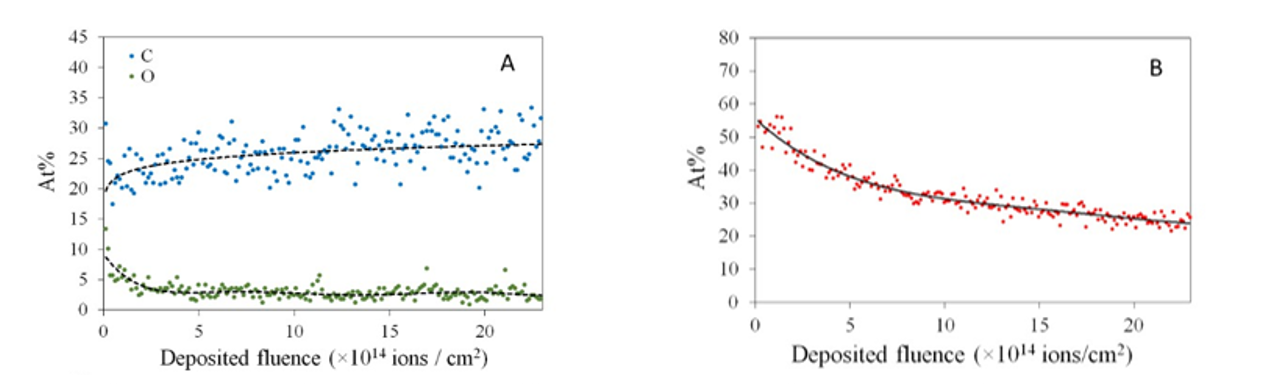
Figure 1. The oxygen and carbon (A) and hydrogen content (B) changes of the PMMA polymer caused by 1.6 MeV He+ ion irradiation, measured by real-time RBS/ERDA. Symbols: concentration of carbon, oxygen and hydrogen evaluated from 200 successively recorded spectra taken on the same spot of the sample with a dose of 0.02 µC. Lines: fitted exponential curves.
Egypt as flux. The detection of Sb by PIXE has indicated that to obtain the transparency of the primary glass Antimony oxide was used as decolourant. This result has also supported the Northern Egyptian origin of the primary glasses. All the coloured Byzantine fragments were made from soda-lime glass, proving that commerce with Egypt, the main source of natron was still active. In colouration yellow pigment based on Pb and Sn (as shown in Fig.2). White, green, blue and dark blue paintings contained Ca, Cu, Fe and Mn, respectively.
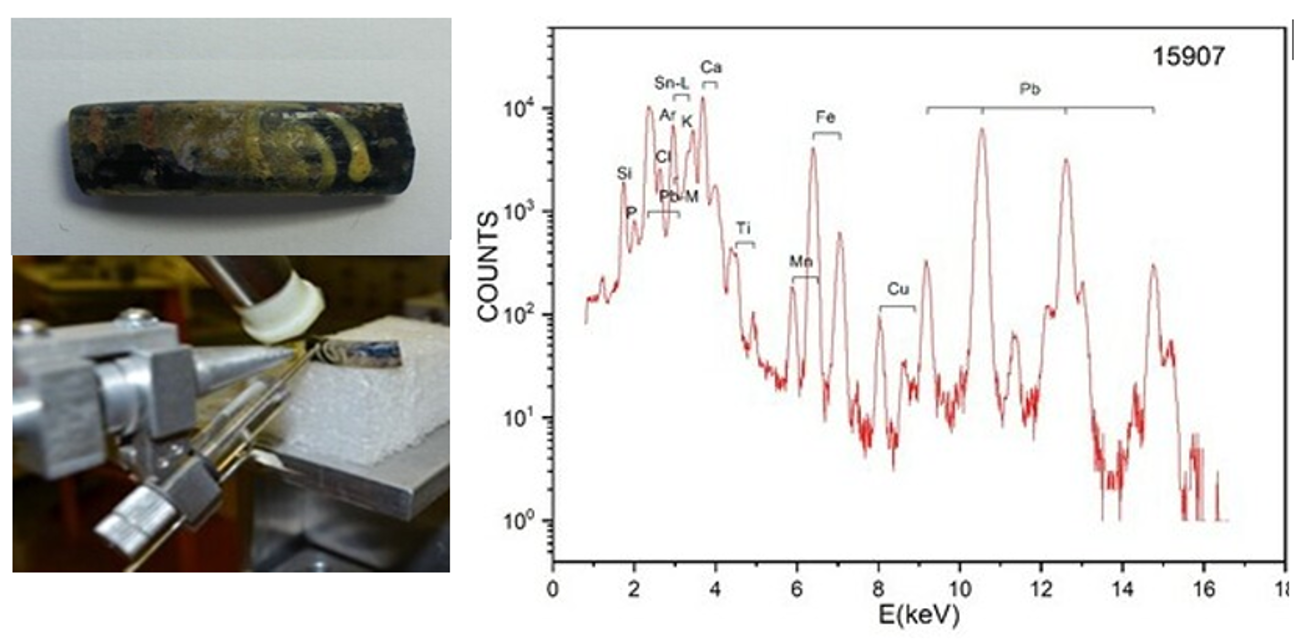
Figure 2. Yellow pigment on a Byzantine bracelet: a combination of Pb and Sn dye.



